
[ To Contents ]
Igor I. Kondrashin
Dialectics of Matter
Dialectical Genesis of Material Systems
(continuation)
The motion of Matter along the coordinate of quality
( goes on with more acceleration (that is at shorter periods of time -)
) in the systems, where the motion in space
( is limited. Owing to this the spatial localisation of fng. units of levels of high organisation, having occurred at a certain stage of the Evolution of material substance as a result of the regrouping of the structure of the Universe into star-planetary formations because of the constancy of the quantity of the aggregate Energy, became the cause of the acceleration of the motion in quality which is confirmed also by the formula)
. One of the hypothetically isolated centres of fnl. evolution of Matter became from some time the star-planetary couple the Sun - the Earth. The principal function of the Sun, as the centre with a predominance of the entropic factor of the system, became: 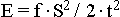
1) the permanent (donor) provider of the whole systemic formation with fng. units of the sublevel AA, a part of which continuously fills in the fnl. cells on the Earth corresponding to them;
2) the replenishment of the microenergetic balance on the Earth because of the possession by the said units of a definite impulse (mV). It is calculated that on all those purposes the Sun expends as a whole about 4 million tons of its mass per every second.
The planet the Earth in its turn is the centre with a predominance of the energetic factor in this bipolar bunch and it serves as an arena for the motion of Matter along the coordinate of quality( at unknown yet in dimensions part of the Universe. Owing to this the subject of our research acquires a more limited space - the surface of the Earth's sphere.)
The development of fng. units of the sublevel E was going on our planet at an early stage of its existence. It is not ruled out that analogous processes are happening as well on the other planets of the Solar system. Nonetheless, starting from the organisational level F, to which the simplest high-molecular compounds are attributed, the description of the systemic processes can be confirmed by the facts only from the history of our planet, as we have no trustworthy information yet about their presence on other planets and we can assume such a possibility only theoretically.
Besides the formation of fng. units of the new level the acceleration of the motion along the coordinate of quality was occurring also owing to a rise of the coefficient of their polyfunctioning. For the systemic organisation of the sublevel F the most useful turned out to be the atoms of carbon C and silicon Si, able because of the peculiarities of their structural construction to make up four chemical connections. If the connections are establishing with fng. units identical to them, then a substance in a solid state is existing only in the form of atomic crystals. The entire volume of such a substance is as if pierced by a thick three-dimensional lattice of atomic links and it is impossible to pick out in it some separate parts - islets, chains or layers.
The most widespread minerals on the surface of the Earth's lithosphere - ordinary and compound silicates - have as the principal construction block an atom of silicon in the tetrahedrons surrounding of four atoms of oxygen. In nature there are three main modifications of the dioxide of silicon (SiO2):
1) quartz, which is thermodynamically steady below 870oC;
2) tridimit, steady from 870oC to 1470oC;
3) crystobalit, steady above 1470oC.
Thus, silicon is one of the most widespread elements in the earth crust. It constitutes 27% of the explored part of the earth crust occupying by prevalence the second place after oxygen. Silicon is the principal element in the compositions of minerals, rocks and soils.
The most widespread element of the earth crust is oxygen. In a free state it is in the atmospheric air, in a bound state it forms part of water, minerals, rocks as well as all organic substances. The total quantity of oxygen in the earth crust is near a half of its mass (about 47%). The natural oxygen consists of three stable isotopes: 16O - (99,76%), 17O - (0,04%) and 18O - (0,2%).
However, the biggest load in the systemic organisation of Matter falls on compounds, a part of which carbon forms. Though its total content in the earth crust is only about 0,1%, by a great number and a variety of its compounds carbon occupies an absolutely particular position among other elements and has the highest coefficient of polyfunctioning among fng. units of the level F. The number of the scrutinised compounds of carbon is estimated nowadays roughly at two million, while compounds of all the other elements, all together, are calculated only by hundreds of thousands. The variety of compounds of carbon is explained by an ability of its atoms to get mixed up in between the formation of lengthy chains or coils.
As it was already noted, by the character of their connections compounds of fng. units are divided into homodesmical and heterodesmical, that serves as one more evidence of the availability of the motion of Matter in quality( . In the case of the existence in nature of only homodesmical connections, that are typical for centres of the energetic factor, the Evolution of Matter would have reached a deadlock, as the structural regrouping of fng. units of the present level would have led to the construction of systems of the level E only with the compact crystal packing. The energy of systems would have volatilized, and the Earth would have turned into a dead stone-metallic globe. The availability of the motion of Matter in quality rules out such a course of events. Therefore the existence of homodesmical systems equally with the action of centres with the entropic factor is conducive to the creation of different high-molecular compounds, each of them bearing this or that new fnl. load additional to the total existing spectrum of functions of the evolving Matter. Functional features of high-molecular compounds first of all are bound with the ability of macromolecules to modify their form without breaking their connections. The mechanism explaining the variety of conformations of macromolecules nowadays is well studied and is widely being used in the chemistry of polymer materials. Therefore we shall not dwell on its description. It is important only to underline here once again that, whatever construction high-molecular compounds would have, whatever their structure would be, we can always define in them invisible fnl. cells and occupying them real fng. units of different sublevels, that is different atoms, molecules, etc. If a fng. unit were to fall out of this or that fnl. cell or fill it in by a fng. unit not corresponding to it this will lead to the destruction of the structure of a given system or to an alteration of its fnl. features.)
In connection with the complexity of their structural construction and the presence of a great number of links all high-molecular compounds exist only in a condensed state - solid or liquid. However, by phase state they correspond more to the structure of liquid, which owing to a high viscosity seems to us in most cases a solid body.
Complex compounds, very various both by the construction and the functional features, constitute a special subgroup of systemic formations of the sublevel F. But in the evolution of the material substance at the present organisational level they play more a secondary, or rather an auxiliary part. Further, at the levels of higher organisation of the material forms, their part is increasing. In particular, such most important natural compounds, determining Life on the Earth, as haemoglobin and chlorophyll, are attributed to intracomplex compounds. The structures of their nuclei are alike, only the fnl. cell of the unit, that initiates the formation of a certain complex, in chlorophyll is occupied by Mg2+, while in haemoglobin by Fe2+. By two vacant coordinational places two more molecules of other substances join easily those units-initiators of complexes occupying the free fnl. cells. So, in haemoglobin from one side of the plate of chelate a molecule of globin protein is connected by ferrum, and from the other side - a molecule of oxygen, owing to which this compound is a carrier of oxygen.
The functional evolution of Matter in the sublevel F and the appearance of new structural formations were and are occurring owing to various transformations of substances by means of the redistribution of electronic densities between the atoms forming them, that leads to the breaking of the preceding and the creation of new intrastructural connections. However, it is enough to remember such chemical transformations as an explosion of gun-powder and the rusting of iron to assert that different structural modifications are moving with quite different velocities - from extremely high to very low. The causes of this are specific peculiarities of every reorganisation, that depend on a balanced spreading of a newly formed structure( in space-time)
( under present conditions as well as the qualitative characteristic of fng. units participating in the reaction.)
Intervals of the duration time of different chemical reactions per a unit of space vary from parts of a second to minutes, hours, days. Some reactions are known to need several years, decades and even longer periods of time for their continuance. If a reaction goes in a homogeneous system, then it is going in the entire volume of this system. As a result of the reaction, as a rule, a heterogeneous system appears:H2SO4 + Na2S2O3 = Na2SO4 + H2O + SO2
+ S
With any monophase mixture, the liquid solution of different substances can serve as examples of a homogeneous system. If a reaction is going between substances, forming a heterogeneous system, then it can go only on the surface of a phase division forming the system. So, for example, a dissolution of a metal in an acid Fe + 2HCl = FeCl2 + H2
can go only on the surface of the metal because it is only here that both reacting substances come into contact one with the other. The result of the reaction is again a heterogeneous system, which under the conditions of lack of locking by means of a dismissal of one of its phases can become a homogeneous system. As examples of heterogeneous systems we can designate the following systems: some water with ice, a saturated solution with sediment, sulphurs in the atmospheric air. At higher stages of the Evolution of Matter as examples of homogeneous systems can be brakes of plants functionally of the same type (a forest, meadow grass, orchards), united groups of animals functionally of the same type (a herd of sheeps, a pack of wolves or monkeys). Heterogeneous systems in this case will be: a herd of horses at a meadow, a team of lumbermen in a forest, production enterprises, etc. Chemical kinetics is engaged in the study of conditions having an influence on velocities of chemical reactions. At higher stages of the Evolution of Matter these problems should be referred to the biological and to the social kinetics accordingly.
The following factors are referred to as the most important, having an influence on velocities of reactions, that go in systems of the level F: functional peculiarities of reacting substances, their concentrations, temperature, the presence of catalysts in a system. Velocities of some heterogeneous reactions depend also on the intensity of the flow of a liquid or a gas near the surface, where a reaction is going. After entering into a reaction of fng. units of two different substances fng. units of a third, a fourth, and etc. substance is being created, which fill in fnl. cells corresponding to them, though theoretically the process is occurring in the opposite order: at first an invisible fnl. cell (C) of a new quality appears, then there is the closing in of obvious fng. units (a and b) and the creation of a new fng. unit (c), which fills in the fnl. cell (C), are going. Therefore velocities of reactions depend on a capacity of reacting substances because of their structural constructions to create new fng. units, that is of spatial locations and mutual connections of initial fng. units of qualitative sublevels, on proportion and quantity of fng. units (a and b) entering into reactions, that is characterised by their concentrations.
Their mutual closing in and collision of one with another (costroke) is the necessary condition so that between particles (molecules, ions) of initial substances a chemical interaction would occur. Speaking precisely, particles should approach each other so much, that atoms of one of them would feel the influence of electrical fields originated by atoms of the other one. Only in such a case would those transitions of electrons and regroupings of atoms become possible, resulting in the formation of molecules of new substances - products of a reaction. However, not every collision of molecules of reacting substances leads to the origination of the product of a reaction. In order that a reaction occurs, that is new molecules form, it is necessary to break or to weaken the connections between the atoms in molecules of initial substances. That requires the spending of some energy. If colliding molecules do not have enough energy, then their collision would not lead to the formation of a molecule: having come into a collision they fly away in different directions like elastic balls.
If the kinetic energy of colliding molecules is enough to weaken or to break the connections, then a collision can initiate a reorganisation of atoms and the formation of a molecule of a new substance. Therefore only those molecules that have a surplus of energy in comparison with the average reserve of energy of all molecules can overcome such an 'energetic barrier' in order to get into a chemical contact with each other. The surplus energy that molecules should have in order that their collision could lead to the formation of a new substance is named the energy of activation of a given reaction. The molecules that have such energy are named active molecules. The surplus energy of those molecules can be forward or rotary for a molecule as a whole, vibratory for atoms, forming it, the energy of excitement for electrons, etc. For each specific reaction only one kind of surplus energy can be principal. With a rise of temperature the number of active molecules is increasing and as a result of that the velocities of chemical reactions are accelerating as well.
The energy of activation of different reactions is different. Its magnitude is the factor by which the influence of reacting substances tells on the velocity of a reaction. For some reactions the energy of activation is insufficient, for others, on the contrary, it is more than enough. If the energy of activation is too insufficient, then it means that most collisions between particles of reacting substances lead to a reaction. The velocity of such a reaction is high. On the contrary, if the energy of activation is more than enough, then it means that only a very small number of collisions of interacting particles leads to a chemical reaction. The velocity of such a reaction is very little.
The reactions, which require some appreciable energy of activation in order to move, start from the breaking or weakening of connections between atoms in molecules of initial substances. During it the substances are getting over into an unsteady intermediate state, which is characterised by a large reserve of energy - an activated complex. Precisely for the formation of which the energy of activation is essential. An unstable activated complex is in existence for a very short time. It is decomposing with the formation of the products of the reaction, during which energy is going out. In a simplest case an activated complex is a configuration of atoms, in which the previous connections are weakened and new ones are being formed. An activated complex arises as an intermediate state during both direct and reverse reaction. Energetically it differs from initial substances by a magnitude of energy of activation of a direct reaction and from final substances - by energy of activation of a reverse reaction. Activation of molecules is possible during the heating or dissolution of a substance, while emitting energy during a reaction itself, while absorbing by them quantums of radiation (light, radio-active, X-ray, etc.), under an effect of supersound or of electrical discharge and even from strokes into sides of a jar.
The velocity of a reaction often depends on the presence in a system of the 'third' component, with which reagents can compose an activated complex. During that an alteration of the velocity of a reaction occurs owing to the alteration of the energy of its activation as intermediate stages of the process would be different. The additional component, which is named a catalyst, after the destruction of the activated complex, does not form part of the products of a reaction, therefore the general equation of the process remains the same. In most cases the effect of a catalyst can be explained by the fact that it reduces the energy of activation of a reaction. In the presence of a catalyst the reaction is going through different intermediate stages, whereas without it, moreover, those stages energetically are more accessible. In other words, in the presence of a catalyst different activated complexes arise, while for their formation less energy is required than during the formation of activated complexes that arise without a catalyst. Thus the energy of activation is going down - some molecules, the energy of which was insufficient for active collisions, now become active.
If a reaction A + BAB is going with a slow velocity, then it is possible to find a substance K, that forms an activated complex with one of the reagents, interacting in its turn with another reagent:
A + B
[A... K]; [A... K] + B
AB + K
If the energy of activation of these stages is lower than the energy of activation of the process in the absence of K, then the total velocity of the process is increasing considerably and such a catalysis is named positive. Otherwise, the velocity of the process would decrease and a catalysis would be negative. Thus a catalyst is a substance that alters the velocity of a reaction and remains after that chemically invariable. A catalyst, present in a system in quantities of a thousand times less than reagents, can alter the velocity of a reaction by hundreds, thousands, millions of times. In certain cases under the effect of catalysts such reactions can be excited, which without them practically do not go on in the given conditions. At the same time, with the help of a catalyst it is possible to alter the velocity only of a thermodynamically possible process. For slowing down undesirable processes or for giving reactions more quiet character negative catalysts are used.
One can discern a homogeneous and a heterogeneous catalysis. In case of a homogeneous catalysis the catalyst and reacting substances form one phase (a gas or a solution). In case of a heterogeneous catalysis the catalyst is in the system in the form of an independent phase and the reaction takes place on its surface.
The catalysis plays a very important part in biological systems. Ferments - plain and complex proteins with big molecular mass - are active catalysts of biological effect. Most of the chemical reactions going on in the digestive system, in blood and cells of animals and men, are catalytic reactions. So, a saliva has the ferment ptyalin, which catalyses the transformation of starch into sugar. The ferment pepsin, present in the stomach, catalyses the desintegration of proteins. Half of an available quantity of urea under ordinary conditions at the temperature 25oC is decomposed by water during 3200 years, but in the presence of the ferment urease the time of its 'half-transformation' at the same temperature is only 10-4 sec. In total more than 30 thousand different ferments are functioning in the organism of a man; each of them serves as an effective catalyst of the corresponding reaction.
On studying heterogeneous reactions, it is not difficult to notice that they are closely linked with the processes of displacement of fng. units of substances, entering a reaction, and new substances. So, to keep the process of the burning of pieces of coal constant it is necessary that dioxide of carbon, forming during this reaction, would be moved away all the time from the surface of the coal and new quantities of oxygen would approach it. Therefore during a heterogeneous reaction one can single out at least three stages:
1) supply of reacting substances;
2) a chemical reaction itself;
3) taking aside the products of the reaction.
The velocity of a chemical reaction, which in its turn can be divided into substages, is determined by the velocity of the slowest substage. A stage, determining the velocity of going of the reaction as a whole, is named the limiting stage. In one case it can be a supply or taking aside substances, in another - a chemical reaction itself.
All chemical reactions are divided into irreversible and reversible. Irreversible reactions are going till the end - until the complete consumption of one of the reacting substances. Reversible reactions are going not till the end: during a reversible reaction no one reacting substance is consumed completely. Consequently an irreversible reaction can go only in one direction, and a reversible one - both in one and in the reverse directions as well. At the beginning of a reversible reaction during the mixture of the initial substances the velocity of the one-direction reaction is high and the velocity of the reverse one is equal to zero. While a reaction is going on the initial substances are being used up and their concentrations are declining. As a result of that the velocity of the one-direction reaction is decreasing. At the same time products of the reaction are being composed and their concentration is increasing. Owing to this the reverse reaction starts going while its velocity gradually grows. When the velocities of the one-direction and the reverse reactions become identical, the chemical (dynamic) balance begins.
By changing the conditions a system is under - concentration of substances, pressure, temperature - it is possible to alter the velocities of the one-direction and the reverse reactions. Then the balance in the system is being broken and moved in the direction of that reaction, the velocity of which became higher. So, during the increase of the concentration of reagents, the velocity of the one-direction reaction naturally is growing and the balance is being moved towards the one-direction reaction, towards more output of products. More output of products can be obtained also by systematically getting them out of the sphere of the reaction, which leads to the decreasing of their concentration in the system and to the deceleration of the reverse reaction in comparison with the one-direction one. For chemical systems, which contain gaseous substances, changes of pressure have the same influence on the shift of the balance as the changes of the concentration of gases. During that the velocity of that reaction is changing more, in which more molecules of gases are participating. The changing of temperature has influence on the displacement of the chemical balance for processes accompanied by thermal effects. If a one-direction reaction is exothermal, then the reverse one is endothermal, and vice versa. For reversible reactions the energy of activation of an endothermal process is more the energy of activation of an exothermal process. In its turn, the more Eact. is, the more the velocity of a reaction depends on temperature. So, an increase of temperature is moving the chemical balance toward an endothermal reaction, as a result of which heat is taken up and the system is cooling down.
On comparing the changes of conditions under which a chemical system is staying with its responding reaction to an outer influence, revealing itself in the moving of the chemical balance, it is not difficult to notice that this reaction always turns out to be opposite to the change of a condition. So, if the concentration of some substance, which is in balance with other reacting substances, is being reduced, then the balance is moving toward the reaction, increasing the concentration of this substance. While increasing the pressure then that process starts going faster, which decreases it, and during the rise in temperature - the process, that causes cooling of the system. These observations form the chemical content of the general principle of behaviour of systems, staying under given conditions in a state of the dynamic balance: if a system, staying in balance, undergoes an influence from without by alteration of some condition, determining the state of balance, then the balance in it is moving toward the process, which leads to the reduction of the effect of the influence. This rule of counteraction is known under the name the principle of La Chattily, formulated by him in 1884.
Thus, for the carrying through of each chemical reaction strictly definite reagents are needed in quantities providing the required going of the reaction under a given temperature and other conditions at a definite velocity, which can be commensurate with temporal intervals. Moreover, every chemical reaction, going under given conditions, has its own definite systemic construction, constituting a combination of fnl. cells which at certain moments are being filled in and set free by fng. units corresponding to them according to the typical for a given reaction algorithm, reflecting the moments of entering the reaction by reagents - fng. units, their possible interchange, while all this is correlated with strictly definite periods of time, fixed by an independent counter of time.
All the simplest and complex molecular compounds of the levels D, E and F are dispersed along the surface of the Earth, and in accordance with their aggregate state form part of the land, oceans and atmosphere of the Earth.
The Evolution of Matter along the sublevel G was going by forming new molecular compounds, which obtained more and more new functions in accordance with the motion of Matter in quality( ).
The differentiation of fnl. cells and formation of new fng. units of the present level were going in the process of the continual combining of fnl. cells of previous sublevels, integrating and modifying their structures, semi-decomposition of these original microsystems to the units of lower sublevels.
The whole process of the Evolution of Matter along the sublevel G has been going for more than 5 billion years in the geospheres of the Earth - spherical covers of different density and composition. For the most part they are atmosphere, hydrosphere and lithosphere (the Earth's crust), which penetrate one into another, are in close interaction, consisting in the exchange of substance and energy, and represent the common system, being pierced by the Sun's radiation.
The outer geosphere is the atmosphere, which in its turn divides into three sub covers: troposphere, stratosphere and ionosphere. Each of these subspheres is characterised by sharply expressed physics peculiarities and bears strictly definite functional loading. The boundaries between these covers are expressed not so sharply and their altitudes are changing both with the time and latitude of a place. The upper boundary of the troposphere is within the bounds from 8 to 18 km. The troposphere unites more than 79% of the mass of atmosphere. The stratosphere is extended till the altitude of about 80 km, constituting approximately 20% of the total mass of the atmosphere. Above the stratosphere is located ionosphere, having less than 0.5% of the total mass of the atmosphere.
The troposphere, where almost all water steam is concentrated, is characterised by almost full transparency with regard to the short-wave sun radiation passing through it, and by considerable absorption of the long-wave (thermal) radiation of the Earth, caused mainly by water steam and clouds. Therefore the troposphere is warming mainly from the earthy surface, as a result of which is the drop of temperature with altitude. In its turn this leads to the vertical mixing of air, the condensation of water steam, and the formation of clouds, rain and snow. The composition of the troposphere includes (by volume) 78.08% of nitrogen; 20.95% of oxygen; 0.93% of argon and about 0.03% of carbonic acid gas. 0.01% consists of hydrogen, neon, helium, krypton, xenon, ammonia, peroxide of hydrogen, iodine and others.
The composition of dry air in the stratosphere differs by a very important peculiarity - by increasing with altitude both the total concentration and relative content of ozone (three-atom oxygen). Ozone is being formed in the stratosphere as a result of the dissociation of molecules of oxygen under the influence of ultra-violet radiation of the Sun and the subsequent joining of the turned out atom of oxygen with another molecule of oxygen. Ozone is located in the atmosphere in the form of a diffused layer, extended from the Earth's surface approximately 60 km. If all the ozone of the atmosphere concentrated in the form of the layer under the overground pressure, then the pellicle with thickness 2 - 3 mm could be seen. Despite so insignificant a quantity the importance of the ozone in the atmosphere is exceptionally great due to the extremely strong absorption by ozone of the radiation of both the Sun and the Earth. So, owing to being absorbed by ozone, the ultra-violet radiation of the Sun almost does not reach the troposphere at all.
The ionosphere, the outer sphere of the atmosphere, gets the diverse radiation of the Sun and stars. Its structure consists mainly of atoms of oxygen and other substances.
Between the atmosphere and the solid stone earth-crust there is an interrupted water cover - the hydrosphere, covering nowadays 70.8% (361 mln. sq. km) of the surface of the Earth. It constitutes the aggregate of oceans, seas and continental water basins. The chemical composition of the hydrosphere is expressed by the following figures: O - 85.82%, H - 10.72%, Cl - 1.9%, Na - 1.05%, Mg - 0.14%, S - 0.088%, Ca - 0.04%, K - 0.038% , etc. The age of the hydrosphere is not less than 2 bln. years. In the hydrosphere Life on Earth was originated for the first time. The evolution of organisms went on here during the whole pre-Cambrian period, and only at the beginning of the Palaeozoic era did animal and vegetable organisms start to move gradually to land. The main component of the hydrosphere is water - one of the most widespread substances on the Earth. A lot of this water is in the gaseous state in the form of steams in the atmosphere; during the whole year it is situated in the form of huge masses of snow and ice on the tops of high mountains and in Arctic regions. In the depths of the Earth there is also water, soaking soil and rocks. Water has rather high coefficient of polyfunctionality and bears a large spectrum of functions to be fulfilled. Being the first cradle of the origin of Life, water in each organism constitutes habitat, in which chemical processes, which provide the vital activity of organisms, take place; moreover it itself participates in a large number of biochemical reactions. In the form of different solutions water carries out the functions of displacement (transportation) of different fng. units from the place of their synthesis to the place of their functioning in the structure of organism. Being a highly reactionary capable substance, water is an active chemical reagent; very often it carries out the functions of a catalyst. Having an anomalously high thermal capacity it serves as a natural thermal accumulator.
The solid body of the Earth has three main geospheres: the nucleus of the Earth, the intermediate cover and the earth-crust. The radius of the nucleus is about 3500 km. The intermediate cover fills the space from the nucleus' surface to the lower surface of the earth-crust and has the thickness of about 2900 km. The earth-crust, or the lithosphere, is the upper solid cover of the Earth with thickness 15 - 70 km; from above it is limited by the atmosphere and the hydrosphere. The earth's crust has a stratified structure, various in different places. The uppermost layer is occupied by sedimentary cover (the stratisphere). It is interrupted, has the depth to 10 - 15 km and consists of sedimentary rocks, among which clays and argillaceous schist predominate. Sands and sandstone, limestone and dolomites constitute its smaller part.
The formation of the stratisphere began in the ancient pre-Cambrian period and lasts until now. The total age of the earth's crust is defined as 3 - 3.5 bln. years, but the age of the most ancient, accessible for observation, pre-Cambrian geological formations rather exceed 2 bln. years. The sedimentary cover was formed as a result of the lengthy process of differentiation of the lithosphere's substance under the influence of tectonic moves, the solar energy and vital activity of organisms. This process was accompanied by a complex interchange of substances between the granite and basaltic covers of the Earth, from one side, and the atmosphere and the hydrosphere, from the other. The chemical composition of the stratisphere together with the salt composition of the ocean is close to the average composition of the earth's crust as a whole.
During the geological history of the Earth natural alterations of the inner structure and consistency of the earth-crust, of the relief of its surface, of the character of outer and inner geological processes were going on. So, for instance, the rocks of the most ancient Archaen era everywhere are much metamorphosed (recrystallised), pierced by intrusions of magma and crumpled into folds. Along the entire surface of continents mountains arose repeatedly, which went to ruins later on. During proterozoa and after that the continents, while going down, were partly flooded with sea and, after getting up, again turned into dry land. Simultaneously powerful moves of the earth-crust went on in different places, as a result of which numerous mountain ranges were arising, later ruined. Contemporary inner geological processes reveal themselves:
1) in slow raising and lowering of the earth's surface at the rate of several centimetres per year in mountainous areas, but the usual rate amounts to some millimetres per year;
2) in abrupt moves of some parts of the earth-crust - earthquakes;
3) in volcanic eruptions.
As a result of the above geological processes and also under the permanent influence of the atmosphere (including the sun and cosmic radiation), the hydrosphere and the biosphere during two bln. years the formation of the principal layer of the lithosphere - the soil - was taking place.
Its formation went on from friable rocks, that is from the fng. units of the sublevels D - F: clays, loam, sandy loam and sands, constituting the products of the weathering of magmatic, metamorphosed or dense sedimentary rocks, deposited at places of their origination or, more often, having undergone transfers and redeposits (often repeated) by fluid water or wind. The soil consists of the firm, liquid (the soil solution) and gaseous (the soil air) parts. In the firm part the principal mass share is usually occupied by the mineral part, represented by small (most of them are from 1 mm to tenth and hundredth parts of micron) particles of different minerals. The composition of soil is formed by the following chemical compounds (in decreasing order): SiO2, Al2O3, Fe2O3, K2O, Na2O, MgO, CaO, CO2, Cl, SO4 and by many others. But the most valuable component of the soil is humus - the final result of the functional development of Matter along the organisational level G. The composition of humus is formed by different high-molecular acids, among which groups of gumming, ulmic and fulvo acids have the greatest importance. Chains of aromatic nuclei of two- and three-member phenols form the basis of complex molecules of gumming acids. Different functional groups are joined to them: carbocsilic, methocsilic, spirituous and others.
All the numerous chemical compounds of the sublevel G, including also humus substances, constitute complex systemic formations, enclosing into its fnl. cells fng. units of all the foregoing sublevels from a to E. Each of these particles, in the form of a certain way of organised structures of Matter, bears at its organisational level different functional loads, that considerably differ from each other. However, as it was already at the previous stages of the Evolution of Matter, each stable systemic formation of the sublevel G at a certain moment becomes a fng. unit of the following organisational level - H (the biosphere). And as soon as the actual point of the invisible line of the tensor of the Evolution of Matter moved from the level G to the level H, immediately the level G remained out of bounds of the sphere of actual development of Matter and became, as also all the foregoing organisational levels, a supplier of functional half-finished products - fng. units of its sublevel - for the formation of functional systems of the level H.
The humus horizon of the soil serves as a natural accumulator of these half-finished products, consisting mainly from its organic substance. Being the very upper layer of the soil and coming into direct contact with the atmosphere and partly with the hydrosphere, the humus horizon has relatively small thickness. It varies in different grounds from several centimetres to one, sometimes to 1.5 m. In areas of deserts, half-deserts, mountains, etc., the humus horizon is practically absent. But even at those places where it is sizeable, the content of humus in the upper part of the humus horizon ranges from tenth parts of a percent to 15 - 18%. Thus the formation, functioning and development of fnl. systems and fng. units of all following organisational levels of Matter depends directly on the quantitative composition of half-finished products being situated in the humus horizon - the accumulator. But as this accumulator for many millions of years has practically an invariable area( , it serves as one of the principal natural regulators of numbers of all living things on the Earth just in the same degree, as all living things on the Earth themselves in order to avoid the worst consequences should self-regulate its numbers in accordance with the resources of this stage of the systemic organisation of Matter.)
[ To Contents ] [ Next ]